Secure Your Email
Encrypt and digitally sign your emails with publicly trusted S/MIME Certificates
- Ensure privacy of sensitive information
- Authenticate email sender to distinguish from phishing attacks
- Create a tamper-evident seal around email contents
Start Securing Your Emails
You need an S/MIME Certificate to digitally sign and encrypt your emails. Pricing depends on the level of verification and the amount of information contained in the certificate
Please ensure to order your certificate from the Region where the company in the certificate is based, and with the correct currency. If your certificate request upon ordering does not match these criteria, your order may risk being delayed, and/or not being processed.
Select RegionDigitally Sign & Encrypt Emails
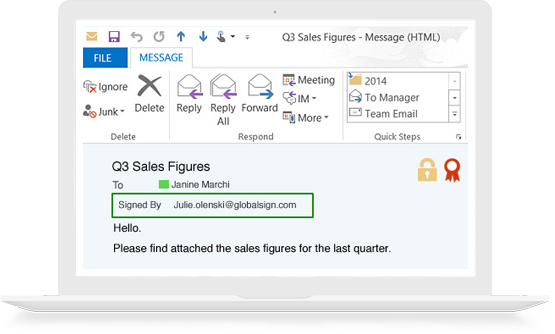
S/MIME Certificates allow you to digitally sign and encrypt emails so you can prove your emails actually came from you and not an imposter, and ensure that only your intended recipient can access the encrypted contents.
Why choose GlobalSign for S/MIME?
Need multiple S/MIME Certificates?
Use our certificate management platform to make your life much easier and save money compared to purchasing separate certificates. Centralizing all certificates simplifies lifecycle management, helps avoid unexpected expirations, and makes it easy to set budgets.
Learn MoreHelpful Resources
- What is S/MIME and how does it work?
- How to spot a phishing email
- Encrypting email vs encrypting servers - what’s the difference?
- How to use S/MIME Certificates in Microsoft Outlook
- How to use S/MIME Certificates in Thunderbird
- How to download and install your S/MIME Certificate
- How to download and install your certificate in Mac OS




